
So hopeful that helped you understand the difference between entropy and enthalpy.\nonumber\]īoth ways to calculate the standard free energy change at 25 ☌ give the same numerical value (to three significant figures), and both predict that the process is nonspontaneous (not spontaneous) at room temperature (since \(ΔG^o > 0\). So that's the difference between delta H and delta S, and it helps understanding why elements have a zero delta H, but not a zero S. We must measure the change in energy or change in heat. H is the measurement of how much energy it contains within it. So delta S is the measure of randomness or chaos or movement, as in the particles or compounds. Outsourced: Cost averages 100 per sample for the first 5 years, increasing to125 per sample for years. There is no significant salvage value for the equipment and supplies currently owned. So it's going from a high disorder to a low disorder. In-house: Equipment and supplies initially cost 125,000 for a life of 8 years, an AOC of15,000, and annual salaries of 175,000. It's actually just saying my delta S is decreasing in value. My delta S is going from high to low, it's going to be negative. So my delta S and then with a numerical value of it. So over here, the products it's all solids. So you have a high amount of disorder over here. So we have, this is a solid state, this is a gaseous state, this is a solid state. How much disorder or chaos is actually happening within the system. So iron and oxygen were high in energy, then it released the energy when it became rust or Fe2O3.ĭelta S, we have to look at the states of matter. So it does require energy or releases energy when this reaction occurs. But this, it does have a delta H and I'm going to say this is the reaction. Formation reactions you're only allowed to have one mole of a product. So this actually is a formation reaction, well not really, because it doesn't have one mole. Iron and oxygen combine together to make rust. What about a process? Well process we use half delta H's. 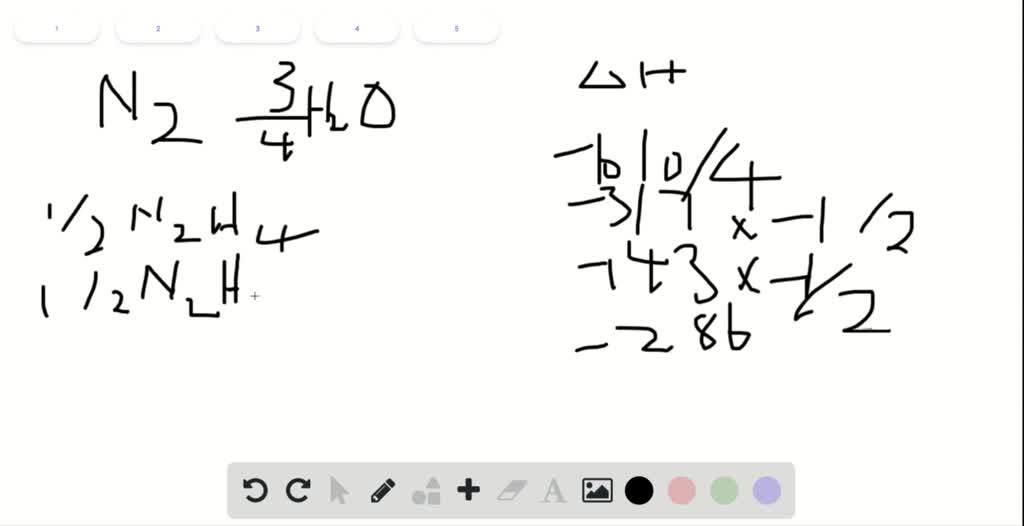
They do have some sort of amount of energy and movement within it. Every substance has measurement of disorder or chaos and movement. So there is no delta H but there is an S, and S is measuring the movement, or the disorder or the chaos in this oxygen has. However oxygen isn't made, therefore it doesn't have the energy, it doesn't need energy to create because it is naturally created. They are constant moving, so they do have mass measurement. They can turn this way, if they want to turn this way. They can squeeze together, they can stretch apart. So oxygen can be together, it can be stretched apart, these bonds are mobile. So it moves in space, so S is the movement, measurement of randomness, disorder, movement. O2 looks like this double-bonded, it's a lot of electrons around it, that's too many. What about delta S? What the heck is that? But since we don't make elements, the delta H for elements is zero. C2H2(g) O2(g) arrow 2CO2(g) H2O(l) Delta H -1299.6 kJ C(s) O2(g) arrow CO2(g) Delta H -393.5 kJ H2(g) O2(g) arrow H2O(l) Delta H -285.8 kJ Calculate Delta Hrxn for the following reaction: CH4(g) 2Cl2(g) arrow CH2Cl2(g) 2HCl(g) Use the following reactions and given Delta Hs. So it's not actually what is in the heat that it actually contains just as compound, it's actually how the compound is made, how much energy it takes to make that compound. For elements it's always zero, even the o2 or the diatomics. It actually is a process going from its elements reforming that. For compounds, when we're talking of the delta H, it's a formation of that. So if we have an element, elements have a delta H of zero, always. So we're going to have elements and compounds. S is the measurement of this disorder and randomness and movement, within a particle or a process. We can however measure S, and this is how we do it. We don't have the means of measuring that. You cannot have this H by itself, we can't measure it. We can only measure the change it undergoes through a chemical process. We cannot decipher how much heat or energy something has in it. Well H is the measurement of heat or energy, but it's a measurement of the transfer of heat or energy. Calculate the equilibrium constant for the reaction. P4O10(s) 6H2O(l) to 4H3PO4(s) The Delta G of the reaction is -7.050 kJ mol-1. 2Mg(s) O2(g) 2MgO(s) Calculate Delta H, Delta S, and Delta G for the following reaction at 25 degC. You will have to look up the thermodynamic information. We can however talk about just straight up H and S, let's do that. Calculate delta G for the reaction at 25oC. That's because we typically talk about changes, reactions or processes that actually happen in Chemistry. 
It's a measurement of randomness or disorder. Now I'm just giving very simplified definitions, but you should have these in your heads.ĭelta S is entropy. This measurement of heat or energy transfer. And the reason I made my H capitalised, is because that's how I remember that delta H is enthalpy. Let's decide what delta H and delta S are. Tips on understanding the difference between delta H and delta S.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
